- Home
- About
- Contact
- Use adobe illustrator cc
- Clean my space 3 sweep
- Some kind of wonderful free online
- List two solutions tarbell suggested to break standard oil-s monopoly
- Nuance dragon naturallyspeaking premium 13 speech recognition software
- Gta 4 lag fix mod gtaall
- Mlb the show 17 walmart
- Minecraft 1-7-1 optimal
- Iso 13485 clauses
- Serato dj 1-9-6 dj plugin flex fx
- Is my office 32bit
- Dragon ball fusions 3ds us release
- Youtube bee gees greatest hits full album
- Kodi 16-1 box channels

Such procedures must be capable of implementation at any time. (Clause 7.5.2.2)ġ0. Identification –to ensure that devices returned are identified and distinguished from conforming product (Clause 7.5.3.1)ġ1. Traceability –to define the extent of product traceability and the records that must be kept (Clause 7.5.3.2.1).ġ2. Preservation of Product – to ensure the conformity of product during internal processing and delivery to the intended destination (Clause 7.5.5).ġ3. Control of Monitoring and Measuring Devices – to ensure that monitoring and measurement can be and is carried out consistently with the monitoring and measurement requirements (Clause 7.6)ġ4. Feedback – to ensure an adequate system to provide early warning of quality problems and for input into the CAR/PAR processes (Clause 8.2.1).ġ5. Analysis of Data – to ensure that data demonstrating the suitability and effectiveness of the quality management system is determined, collected and analyzed and to evaluate any improvements to QMS effectiveness (Clause 8.4).ġ6. Advisory Notices: Improvement, General – to address the issuance and implementation of advisory notices and notification to regulatory authorities, as required.
ISO 13485 CLAUSES ISO
In addition, the following standard sections of ISO 13485:2016 require ‘the establishment of 10 mandatory documented procedures in addition to ISO 9001:2008’ to define and control the requirements and practices of these operational areas:ħ.     Design & Development – to ensure a controlled, consistent design and development process (Clause 7.3.1)Ĩ.     Purchasing Process –to ensure the purchased product conforms to specified purchase requirements (Clause 7.4.1)ĩ.     Particular Requirements for Sterile Medical Devices – to ensure validation of sterilization processes.
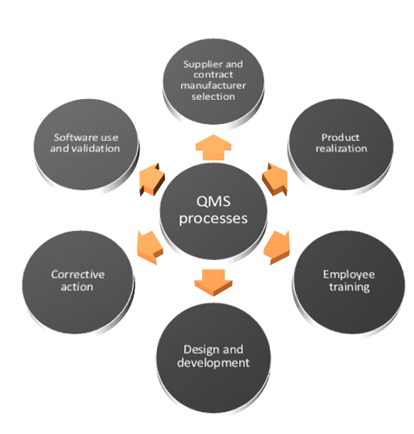
Please refer the newsletter of the PROCERT UK Benefit from the positive effects of a quality management system: lean processes, efficient work flows, and international recognition.

We examine your quality management practices for medical devices, focusing on design and development, production, customer service, and assembly. EN ISO 13485 is the international standard for quality management systems for medical devices. Validating Your Quality Management SystemĬertifications build trust – a crucial factor when it comes to medical devices.  ISO 13485 Certification is an objective evidence of compliance of the requirements

Exporting medical Devices to various countries is mandatory. The ISO 13485 standard is for medical devices – quality management systems – requirements for regulatory purposes, is the basis for regulatory compliance in local and most global markets. ISO 13485 Standard has been based on the requirements of ISO 9001 standard and has the same structure in terms of clauses. Even some clauses of ISO 13485 have been referred to ISO 9001. The main objective of the standard is to facilitate harmonized quality management system requirements for regulatory purposes within the medical devices sector. ISO 13485 is an internationally published standard that defines requirements within quality management systems for manufacturers, suppliers, contract service and distributors of medical devices and equipment.
